A public service message from FDA Intern:
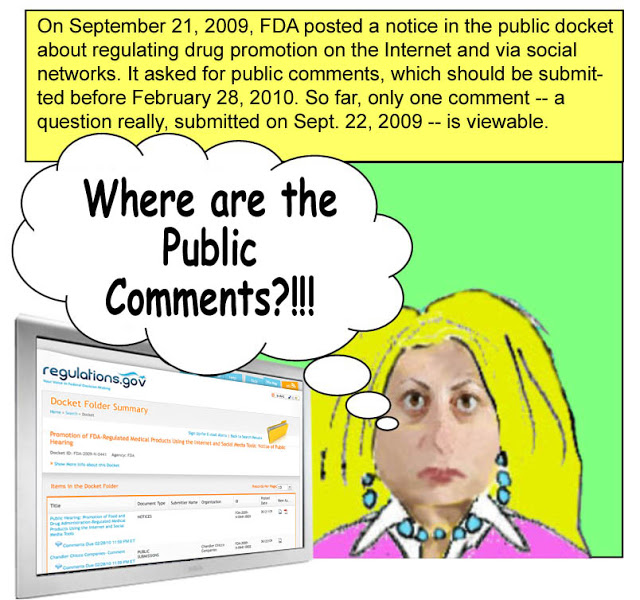
Not only does FDA Intern want to know where all the comments are, I want to know also! It’s been over 2 months since the FDA called for comments and three weeks since the public hearing. But there are NO comments available to view on the www.regulations.gov docket here. There is ONE question that was submitted on September 22, 2009, but no comments.
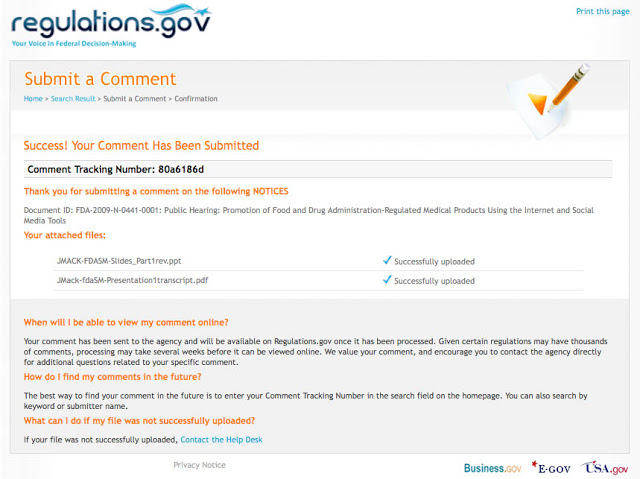
I submitted a comment this morning. It was the script and PowerPoint deck of the presentation I made during the first day of the public hearing. Here’s the confirmation screen:
My comment is not yet publicly available. When you submit a comment, you are warned that it may take “several weeks” for it to be available in the public docket. Why is that? I notice that Docket: FDA-2009-N-0294 (Regulation of Tobacco Products; Request for Comments) has over 800 comments publicly available — the first one was submitted 2 days after the notice was published on July 1, 2009. More importantly, the last comment was submitted YESTERDAY and is now publicly viewable!
So, where are the public comments for Docket FDA-2009-N-0441?
Having comments publicly available as soon as possible should be part of FDA’s Transparency Task Force.
More Questions
When will comments be made public is just one of the MANY questions stakeholders want to ask the FDA about the process following the public hearing. I have posed a few others in the “What Happens Now?” survey I started on 19 November 2009 (access it online here). You can pose your own question and/or chose one or more of the following as the questions you’d like answered by the FDA:
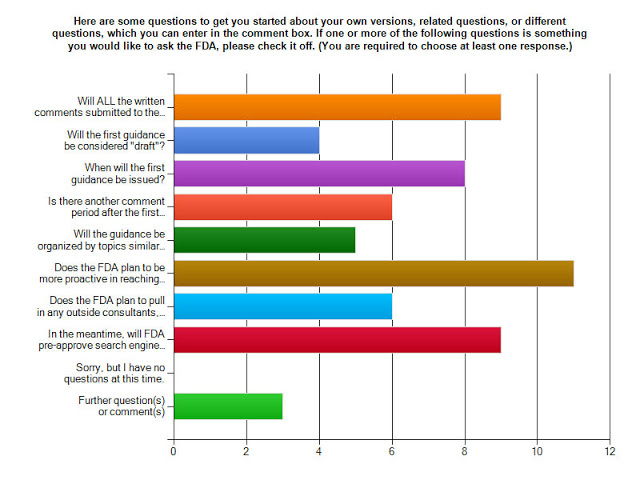
- Will ALL the written comments submitted to the docket be made available to the public for viewing?
- Will the first guidance be considered “draft”?
- When will the first guidance be issued?
- Is there another comment period after the first (draft) guidance is issued?
- Will the guidance be organized by topics similar to how the docket was structured?
- Does the FDA plan to be more proactive in reaching out to more stakeholders — patients, public, and physicians in particular — during the comment period?
- If so, How will it do that?
- What can we do to help?
- Does the FDA plan to pull in any outside consultants, or hire additional experts internally, to help craft the guidelines?
- In the meantime, will FDA pre-approve search engine ads, especially ads using Google’s new format?
So far, here’s how respondents voted:
Fabio Gratton, Chief Innovation Officer at Ignite Health, has been diligently working to collect a list of questions for the FDA based upon responses to my survey and collected by him here, where he is organizing them into categories.
Once Fabio and I have collected a thorough list, we will send it to the FDA. Realistically, the Agency might not be able to answer all the questions, but we believe any answer will provide us with more insight than we have now. It certainly will help FDA understand our concerns.
Postscript:
I just received an e-mail response from Jean-Ah Kang, Special Assistant to the Director, DDMAC – CDER, in response to this post. “FYI,” said Jean-Ah, “we are working with our counterparts in the Division of Dockets Management to ensure that all comments are posted for public view.”