 Long before there was a Hanna Montana there was a Wanda Nevada — a 1979 movie starring Brooke Shields and Peter Fonda. I saw the movie yesterday.
Long before there was a Hanna Montana there was a Wanda Nevada — a 1979 movie starring Brooke Shields and Peter Fonda. I saw the movie yesterday.
I also saw the first TV ad for Latisse starring Brooke Shields last night during a re-run of “Desperate Housewives.” Latisse is Allergan’s “new” drug approved by the FDA for “hypotrichosis.”
Hypotrichosis, according to a Wikipedia entry, “is the term dermatologists use to describe a condition of no hair growth. Unlike alopecia, which describes hair loss where formerly there was hair growth, hypotrichosis describes a situation where there wasn’t any hair growth in the first place.”
That, however, is NOT how Allergan describes the condition. According to the “Patient Information” sheet, “Hypotrichosis is another name for having inadequate or not enough eyelashes.”
I imagine asking asking a woman if she has “adequate” or “enough” eyelashes is like asking a man if he has a “big” enough or “hard” enough penis. It is unlikely, therefore, that any woman wouldn’t want, at least, to try this product once.
BTW, the “full prescribing information,” which I am sure nary a single consumer will ever read, says “LATISSE™ (bimatoprost ophthalmic solution) 0.03% is indicated to treat hypotrichosis of the eyelashes by increasing their growth including length, thickness and darkness.” Nothing about “inadequacy” or “not enough” here.
In the TV ad and Latisse Web site, Brooke explains why she was a good candidate for Latisse:
“I thought I’d be a good candidate for Latisse,” says Brooke in her video diary on the Web site, “simply because over the years I’ve just been ripping off my false eyelashes while on Broadway and Allergan approached me and said ‘this is a product, it works, it’s FDA approved,’ and my interest just piqued!”
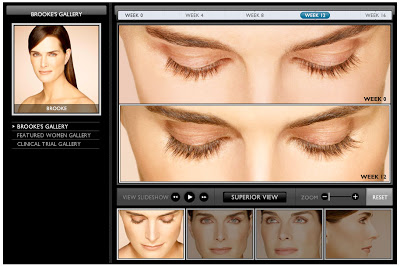
I’m sure it “piqued” (or should I say “peaked”) even more when Allergan told Brooke how much they were willing to pay her to shill their product! Watch her interest “peak” in the video here. You can also amuse yourself in “Brooke’s Gallery,” where you can see “before” and “after” photos — over a 16-week period — from every possible angle and zoom in and out! From the looks of it, Brooke earned every penny Allergan paid her! I wonder if the product manager got a personalized and autographed photo?
 Brooke, like many users of Rx medications before her, is impressed that her medication is “FDA approved.” As if that is equivalent to saying it is completely safe.
Brooke, like many users of Rx medications before her, is impressed that her medication is “FDA approved.” As if that is equivalent to saying it is completely safe.
Recently, the FDA came out with some draft guidance (see “Communicating Risk in Online Drug Ads: Reading the Tea Leaves in Recent FDA Draft Guidance“) that indicates it may have a problem with patients underestimating the side effects of drugs just because FDA has approved them or approved the ads (or statements from drug companies) about them. “Consumers have preconceived ideas about the amount of scrutiny these ads undergo. Many believe FDA exercises tight regulatory control over the content of these ads and to some extent, believe that all ads have been pre-reviewed prior to airing. As a result, consumers are likely to expect that the most relevant risks have been included in the ad.”
Perhaps Brooke is not aware of one the possible “relevant risk” that’s mentioned in the “About Safety” page of the Latisse web site:
“Increased brown iris pigmentation has occurred when similar medications were instilled directly into the eye to treat elevated intraocular pressure/glaucoma. Although iris pigmentation was not reported in clinical studies with LATISSE™, patients should be advised about the potential for increased brown iris pigmentation which is likely to be permanent.”
I have commented on this possible side effect back in October, 2008: “Allergan’s Secret Plan to Thwart Homeland Security and the FDA Approval Process.“
When Allergan says “similar medications,” it really should have said “this medication” because Latisse is just another name for Lumigan, Allergan’s anti-glacoma drug. Both are (bimatoprost ophthalmic solution) 0.03%.
Of course, application of Latisse is outside the eye, whereas Lumigan is applied to the eye. Yet, accidents happen and some Latisse can get into the eye if not carefully applied.
It would be a shame if Brooke’s blue eyes turned brown.














Interesting article about Latisse. I've been hearing a lot about it recently. I use an OTC product called Revitalash, created by an eye Doctor. I got the results I hoped for, though it took about 8-9 weeks. Latisse sounds scary!! And–expensive! The fact that it may change eyecolor is enough to keep me away. I'll stick with what works (on my lashes and wallet!!)
The results don't seem any better than what most women will get with a couple of coats of Maybelline.
Yet another case of manufacturing a condition in order to boost sales.
PLEASE read this: I used Latisse, which was prescribed my my eye specialist for Glaucoma. Nowhere in the side effects did it say that one of it's effects was lash growth where it was NOT supposed to be! I had eyelashes growing in my tear ducts and around that area. They had to be pulled out one by one with tweezers! It was SO painful since NO medication could be used. My pharmacist contacted the manufacturer of this drug to let them know that this was NOT stated in the possible side effects sections. They did NOT respond!!!!Someone Has to let Brooke Shields know what she is recommending with her ads for this product. Or does she know and NOT CARE!!!
"It would be a shame if Brooke's blue eyes turned brown." A shame? And what exactly is wrong with brown eyes?
She just wouldn't be herself anymore. How would she feel?
"A shame? And what exactly is wrong with brown eyes?"
Everything, blue eyes look better.
Latisse would not be prescribed for glaucoma, so either you're not telling the truth, you're confused, or you need to sue your 'eye specialist.'
If you read the post carefully, you will see that Latisse, under a different name (Lumigan), is indicated for glacoma:
"Latisse is just another name for Lumigan, Allergan's anti-glacoma drug. Both are (bimatoprost ophthalmic solution) 0.03%."
You actually can get growth in the tear-duct area of the eye if TOO MUCH product is applied. One drop per eye is all you need and it shouldn't be applied that far down. Latisse causes lashes to grow longer, thicker and darker. And it totally works! If you dont want that to happen in the tear-duct area, do not apply it there. If your product drips, you are using too much.
one's interest is "piqued" not "peaked"
Thanks, Mary. I made the correction.
Before I changed careers I worked for many years in the health care field. When I first learned of Latisse I was intrigued. Sounded like a dream product. But, of course, I knew I had to do the research first. A training I've never let go of. But even for me this information seemed hard to find…or rather, not as easy to pin down as it should have been.
These are the questions I had, after surfing for answers (the lack of detailed clarity on 'medical' websites was absurd):
1) An 'increase' in the brown pigmentation of the colored part of the eye, meaning the iris? If so, it implies the eye would already have to have some amount of the color brown – and that it *increases* the amount or intensity of the brown color. So does this mean that blue eyes, for instance would not be affected at all? Or is "increase," which is what many websites are saying, not the right word? Does it potentially cause blue eyes to irreversibly become brown – in full or in part?
2) Why does it grow hair on other parts of the body? No matter where Latisse is applied hair can grow on a random body part? Or does it only grow where there has been direct contact with the product?
3) Why is Bimatoprost part of the formula? How does it contribute to the intended result of more/longer eyelashes? Seems like it might be an unnecessary risk to include it unless there is definite rationale.
——–
Thanks for posting this information! That it's only a new label for another medication is important to understand. What you have here is more forthcoming than 'medical' the websites I've scoured that were supposedly disclosing 'helpful' information about "Latisse." Only a website about the FDA and legal concerns re: this treatment seemed to be pointing consumers to more of the truth. This is not anything anyone should have to dig for. Also, I'm surprised at some of the comments! Clearly your post was not thoroughly read by all. And of course any liquid dropped onto or into the eye may find it's way into the tear duct, as one person posting insisted should not happen. What could be the motivation behind such criticism of another person's experience?
This is a good contribution, John Mack. Your blog is saved to favorites.
Brooke has green eyes, not blue eyes!
Whatever.
You can't turn non brown eyes to brown. Please read the following:
…This has only happened with brown eyes, never in blue or green eyes, because lack of pigment.
http://www.greenwichdermatology.com/sscms/latisse-2/
No pigment=no brown.
They aren't saying there is anything wrong with brown eyes, just wouldn't it be a shame that in someones quest for something as silly as longer eyelashes, it actually changed the natural color of their eyes.
My eyes were blue green and are now on the border of brown. I watch my other color fade away every day even months after I stopped using Latisse. I hope Brooke's eyes turn brown so she can experience the identity crisis that others are going through. It was so irresponsible for her to be a spokeswoman for this. I hate that product and the greedy people behind it. I also now have dry eyes an eye pressure. Thanks for making me so beautiful Latisse…. you really worked wonders!
liar.
shame on you, liar.
I am 53 years old and have never had long or full eyelashes. I started using Latisse just before Thanksgiving. My lashes are now twice as long as before. They now have a curl to them so I don't have to use a eyelash curler. Before I started using Latisse I had a small area where I did have any lashes at all and that has now filled in.
I have blue eyes and they are still blue. I am very glad that I gave Latisse a try. It certainly has worked for me.
how do blue eyes look better excatly? … :S
I did not say brown eyes "look better" than blue eyes. I said it would be a shame if Brooke's eyes turned brown — because here career, of one thing, depends on her blue/green eyes. Green/blue might be better for her. If, as people say, her eyes are really green AND Latisse has not effect on green eyes, then it's a moot point.
no somebody said "A shame? And what exactly is wrong with brown eyes?"
Everything, blue eyes look better.
1:03 PM thats what i meant by whats wrong with brown eyes :L
No its just that someone said "A shame? And what exactly is wrong with brown eyes?"
Everything, blue eyes look better.
Thats a load of crap
My dermatologist told me that I wouldn't be a good candidate for this product because I have green in my eyes. I have very blue eyes, but if you look very closely (and I had never before this visit to the derm.) I have a few specks of green in them. He said that this product is not recommended for anyone with any green in their eyes. I was confused when I saw the commercial and saw that Brooke's eyes were in fact, green.
I was advised by doctors of the possibility of the iris color change but my aesthetician went through a "training" and was told that only people with green eyes are in jeapordy. I am currently using Latisse and am nervous about turning my blue eyes brown.
Opinion?
I have light brown eyes and now have speckles of dark brown, about 3 spots in each eye and dark around the iris. I stopped using it, but i must say it does work wonders with the eyelashes. I will miss them 🙁
My mother is 68 and always had short and thin lashes. Her lashes would go in all different directions. She started using Latisse about 1 year ago. She has been very happy, her lashes are longer, fuller, and darker. Her lashes now grow up and curl. However, she already has brown eyes. So I can not say anything about the iris changing color.
Hi, my doctor pulled the plug on my latisse after my aqua colored eyes had strange changes. My left eye had less eye lashes and so I always used more latisse on it. My doctor could not believe it the other day when he did a routine checkup on me. My left eye was completely brown while my right eye was still mostly blue. How could this happen!? How long till my eyes stop looking my a mangy mutt?
Cindy
(And oh why would you want brown eyes anyways? Blue eyes are the c@ts @ss!)
I was really skeptical when I started using Latisse. My friend gave it to me and since I have short eyelashes I was definitely curious. My boyfriend and my mom both advised against it because of the side effects. I used it anyway and after about a month I saw results, after 2 months my boyfriend saw the results and he's getting me my next refill. One bottle definitely can last 2 months. I applied it every other night or so. My eyes are blue and there was no pigmentation change which I pretty much knew wasn't going to happen. I really love the results. I feel like I'm wearing mascara without any and because of this I wear less makeup which just makes me feel better. Honestly, there's a risk for everything nowadays. I'd cut back on my cell phone and microwave usage before i stopped using this.
Brown eyes are ugly.
So plain and boring.
Crap eyes lmao.
That's you're opinion. To me blue eyes are pale, cold, and dead. Especially on a pale complexion. Brown eyes are warm, exotic, and charming. Of course, that's my opinion.
What is wrong with you people? This is a good article and its great to hear real experiences! And yet somehow that turned into an argument about which is better blue eyes or brown eyes… Who cares? Not everyone has the same color and not everyone likes the same color. Give it up! Anyway it would be a shame if I turned my brown eyes blue/ blue eyes brown. Its one thing to choose to change something about yourself… Another to "accidently" do it to yourself. That would be a SHAME! ( just to be real clear to all the people looking for a fight, I do not mean to offend or insult anyone). 🙂
Finally someone who says something of intelligence! Why do you all argue about eye color. I love that we all have different colors. I thought this was supposed to be about Latisse. I am interested in if it will change my eye color. My color varies. They have blue, grey, green on outside with yellow center. Just curious to find out if they will change. I don't want them to. I love my eye color very unique.
Anyone using Latisse is a sucker for a re marketed drug using side effects as eye lash growth for new sales. Using a drug for vanity purpose and market sales profit? this is a reminder of how women are marketing targets, gullible and duped on a daily basis by Pharma!
http://bipolarsoupkitchen-stephany.blogspot.com/2009/10/vanity-poses-health-risk-eyelash-growth.html
i would actually love to turn my eyes brown,,, brown is very sexy and seductive, no other color is a beautiful
Brown eyes are soulful, not cold & steely like blue. Think of Anne Hathaway, Gwen Stephani, Monica Bellucci, Lucy Lu, Cindy Crawford, Ali Landry, Hallie Berry, Beonce Knowls, Salma Hayek, Kim Kardashian, & Demi More among many other sexy women. Don't be jealous…
Thank you for this blog – Could you please answer the questions that Miri had. I actually was wondering about the "increase" in brown pigmentation. I have green eyes and blonde hair, and therefore would love to have dark lashes but I don't want to alter my eye color. I think it is very misleading of Latisse to have Brook Shields pushing the product with green eyes if there is a chance that they may be permanently altered. Are there any indications that this change in pigmentation may happen or is it just a shot in the dark?
Somebody countered that green eyes were not affected. As with any medication, it's best to ask your doctor about any medications he/she prescribes for your use.
My eye doctor gave me a bag full of this stuff after my catarac surgery and it did indeed make eyelashes grow! Inside my eye lids! Had to go to eye doctor to have them removed. Boy, was it painful! People, please do not use this product! It is dangerous and should be recalled.
I was considered by most people to be a strikingly beautiful baby, child and teenager. Many strangers would come up to me and my parents in public places and say what a gorgeous , beautiful baby and child. I never had big lips, I had /have normal size lips and a pretty delicate shaped mouth, and even as a baby and child I had high cheek bones, a small pretty shaped nose, an oval shaped face, very big dark brown beautiful deepset doe eyes ,(which many people mistakingly thought were black because of the contrast of my light red hair and skin) light skin, strawberries and cream complexion, no freckles and red blonde hair that was much redder than strawberry blonde.
My mother was an artist and she went to art school and she even sold some of her sculptures in a few local galleries. She drew me in pastels when I was 5, she sculpted my face when I was 7 and she drew me in charcoal pencil which I sat for when I was 18.
She never drew or sculpted my sister who is 4 years older though and was only considered very cute and average pretty and who looks nothing like me.My sister has green eyes,medium brown hair and doesn't have light skin like me.
My mother saw people feature by feature as an artist and I once asked her if she thought a soap actress was pretty and she said, no she has an ugly nose, and she did, her nose was kind of flat and kind of spread out shaped.
Several make up artists have said I have very beautiful eyes,and even a medical assistant recently said this to me.Chocolate is dark brown,and most people like chocolate a lot,it smells delicious,looks delicious and especially tastes delicious!Nobody thinks about cr*p or mud when they see,smell and taste chocolate even though they are the same colors!
Coffee is also dark brown and a lot of people like it too.And as Van Morrison wrote his song,Brown Eyed Girl,and Bachman Turner Overdrive's song,You Aint Seen Nothing Yet,they sing the line about,and then she looked at me with her big brown eyes.
Oh, and even though quite a few sites are reporting that Brooke's eyes are green,they usually look blue in all her pictures since she was a child and on the Brooke Shields fan site,The Brooke Nook it has a section of her childhood pictures from the 1970's which includes baby pictures from the late 1960's and it has a picture of her with some type of modeling form,and her name and birth date are typed on it,and she looks about 2 or 3,and it says she had blonde hair(it turned brown later in her childhood) and *blue* eyes!
blondes look better with brown eyes (jessica simpson) and brunettes look better with lighter eyes….liz taylor and meg fox……the end.
Oh my gosh… I kind of want Latisse just to make my eyes brown. My lashes are actually just a normal length, or decent, I don't really know (my mom thinks they are long though).
Everyone in my family has green eyes, mine are kinda bluish grey and with my medium brown hair I think brown eyes would be awesome. =/ I know my eye doctor wouldn't prescribe it to me though. I wonder what I would look like with brown eyes.. Maybe they would just turn light brown which would be awesome. The darker my hair gets (I was blonde when younger but getting to dark ash brown now) the stranger my eyes look. I would love to have dark eyes and look defined instead of faded and blind… gaah. But people always say I should be happy with my blue eyes..
Wow, "anonymous" surely speaks highly of herself. She sounds like she suffers from a borderline narcissistic disorder. Wouldn't all of us love to have "doe eyes", "high cheek bones", the "perfect nose" blah, blah, blah. She seems inclined to let us know she has these features. A person with confidence exercises a certain amount of humility, clearly anonymous lacks confidence and requires others to validate her existence otherwise she feels insignificant. Oh, and if she feels like I'm attacking her, then what was the point of her post; she says nothing about the product, her experiences with it, nor replies to anyone else's posts.
Lol I couldn't even read that looooooong vanity post. I skimmed it looking for something a bout latisse, but NOTHING.
Who cares if you had doe freaking eyes and high cheek bones and how your mom always wanted to draw you or whatever what does this have to do with this article your just here to make yourself feel better and/or show off
Or maybe she is actually short and obese and wildly unattractive and feels the need to live vicariously through the stories she tells to invisible strangers who might believe her of the life she wished she'd lived…