UPDATED 4 Jan 2016: FDA approved 45 new drugs in 2015, four more than in 2014 and the highest number since 1996. Twenty-one (21) of those (47%) were “orphan” drugs (find FDA data here). This compares to 17 orphan drugs approved in 2014 or 41% of the total (see CDER New Drug Review: 2015 Update and “2014 Was a Good Year for FDA & Pharma“).
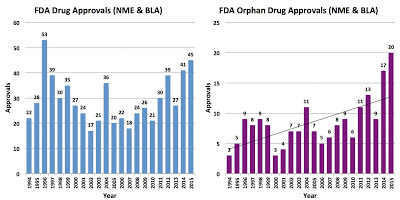 |
| Click on image for an enlarged view. |
That’s good news for the pharma industry, which often submits drugs to the FDA as orphan drugs but once approved the drugs are “used broadly off-label with the lucrative orphan drug protections and exclusivity benefits,” according to the authors of a study recently published in the American Journal of Clinical Oncology (read “#Pharma Welfare: ‘Orphan’ Blockbuster Drugs on Rise – Including Crestor!“).
Pharma marketers also have good news regarding FDA marketing enforcement actions in 2015.
FDA sent out only 9 advertising enforcement letters (2 Warning Letters, 7 Untitled Letters) in 2015, which beats last year’s record low of 10 letters. That was AFTER it “yanked” a warning letter to Pacira Pharmaceuticals about off-label promotion. It apparently did that to avoid a lawsuit filed by the company that claimed the FDA’s regulation violated its First Amendment right (read “Strike 2! FDA Avoids Another Off-Label Court Fight by Capitulating to Pacira’s Demands“).
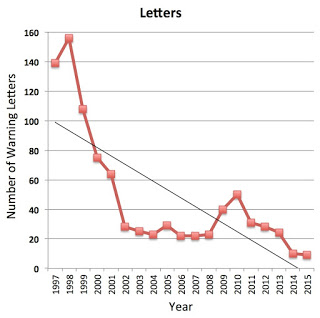 |
| Click on image for an enlarged view. |
“Our success is … measured in our speed and efficiency of approvals,” notes FDA acting Commissioner Stephen M. Ostroff, M.D. in an FDA blog post (here). “The U.S. continues to lead the world in approving novel drugs first. And we’ve seen important progress in our device review program. Our average time to reach decisions on PMAs has dropped 36 percent since 2009.”
Meanwhile, FDA is slow to approve generic drugs, which helps boost drug prices (here).














